Published: 23 November 2025
Last Updated: 10 April 2026
This article is periodically reviewed and updated to reflect current scientific understanding by Vic George.

What Is Iron and Why Is This Essential Mineral Critical for Energy, Oxygen Transport, and Healthy Blood Formation?
Iron is an essential mineral required for transporting oxygen in the blood, generating cellular energy, and supporting immune and cognitive function. In foods, iron appears as heme and non-heme forms, with plant-based non-heme iron (Fe²⁺/Fe³⁺) being readily absorbed when paired with vitamin C. Adequate iron intake is vital for vitality, endurance, and overall metabolic health.
Definition:
Iron is a biologically essential mineral found in legumes, whole grains, leafy greens, seeds, and fortified foods. It forms part of hemoglobin, myoglobin, and numerous enzymes involved in energy production and detoxification. Plant-based diets provide non-heme iron, which is safe, versatile, and absorbable with proper dietary combinations.
Iron exists in two nutritional oxidation states: Fe²⁺ (ferrous iron) and Fe³⁺ (ferric iron). Both contribute to hemoglobin synthesis, oxygen delivery, and mitochondrial energy production. Non-heme iron from plant foods absorbs more slowly than heme iron, giving the body tighter control and reducing oxidative stress. Vitamin C, organic acids, and certain amino acids enhance absorption, while compounds such as phytates or tannins may reduce it. Iron also plays roles in brain development, enzyme activity, and immune resilience.
Key Facts:
- Chemical symbol: Fe
- Mineral class: Essential mineral
- Typical ionic forms in food: Fe²⁺ (ferrous), Fe³⁺ (ferric)
- Key physiological roles: Oxygen transport, energy production, immune function
- Systems supported: Circulatory, muscular, immune, neurological
- Preferred sources: Legumes, leafy greens, seeds, nuts, whole grains, fortified foods
- Deficiency considerations: Fatigue, breathlessness, low stamina, weakened immunity
- Interactions: Vitamin C increases absorption; works with copper, B12, folate in red blood cell formation
Key Takeaways
- Iron is the essential mineral that enables oxygen transport throughout your body, directly impacting your energy levels, cognitive function, and overall vitality.
- Your body absorbs heme iron (from animal sources) up to three times more efficiently than non-heme iron (from plant sources).
- Iron deficiency affects over 2 billion people worldwide and can lead to fatigue, weakened immunity, and impaired physical performance.
- Pairing iron-rich foods with vitamin C sources can increase absorption by up to 300%, making strategic food combinations essential for optimal iron status.
- While iron supplements can help address deficiencies, they should be taken under guidance as excess iron can create harmful oxidative stress in the body.
Iron sits at the core of your body’s energy production system. Without this mineral, oxygen can’t reach your tissues, and cellular energy grinds to a halt. Yet many people struggle with low iron levels without realizing how profoundly it affects their daily vitality.
Nearly every cell in your body depends on iron to function properly. From keeping your mind sharp to maintaining your immune defenses, iron’s oxygen-carrying capability makes it truly irreplaceable. Pure Wellness Solutions provides comprehensive testing and customized supplementation protocols that can help identify and address iron deficiencies before they impact your quality of life.
Iron: Your Body’s Oxygen Delivery System
Think of iron as the ultimate delivery service for oxygen in your body. At the molecular level, iron forms the central component of hemoglobin—the protein in your red blood cells responsible for capturing oxygen in your lungs and releasing it to tissues throughout your body. Without sufficient iron, this oxygen transport system breaks down, leaving cells starved for the oxygen they need to produce energy.
Beyond hemoglobin, iron also forms the core of myoglobin, a similar protein found in your muscle tissues. Myoglobin acts as an oxygen reservoir within muscles, ensuring they have enough oxygen during periods of intense activity. This oxygen storage becomes particularly crucial during exercise when your muscles need rapid access to oxygen to sustain their performance.
The iron-oxygen relationship extends further into your cells through cytochromes and iron-sulfur proteins—essential components of the cellular respiration process. These iron-containing enzymes facilitate the controlled reactions that transform the food you eat into the energy your body uses for everything from thinking to moving.
Why Iron Is Essential for Your Daily Energy
Every single cell in your body requires energy to function, and iron plays a starring role in this energy production process. When you consume food, the nutrients need to be converted into ATP (adenosine triphosphate)—your body’s energy currency. This conversion depends heavily on iron-containing enzymes that facilitate the electron transport chain in your mitochondria, the powerhouses of your cells.
How Iron Creates Cellular Energy
Iron enables cellular energy production through several critical pathways. In the mitochondria, iron-containing enzymes facilitate the electron transport chain—the final and most productive stage of cellular respiration. This process generates approximately 90% of the ATP your body creates, making iron indispensable for energy production.
When oxygen binds to the iron in hemoglobin, it forms oxyhemoglobin, which gives oxygenated blood its bright red color. As this oxygen-rich blood circulates to tissues throughout your body, the oxygen detaches from hemoglobin and diffuses into cells where it’s used in the final stages of cellular respiration. Without sufficient iron, this oxygen delivery system falters, and your energy production suffers accordingly.
The relationship between iron and energy becomes particularly evident during physical activity. As exercise intensity increases, so does your oxygen demand, placing greater reliance on your iron-dependent oxygen transport system. Athletes and highly active individuals often require more iron precisely because their energy and oxygen demands exceed those of more sedentary individuals.
Iron’s Energy Impact by the Numbers: A mere 10% reduction in iron status can decrease aerobic capacity by up to 30% and reduce work performance by 15-30%. This dramatic impact highlights why maintaining optimal iron levels is crucial for anyone seeking to maximize their energy and performance.
The Link Between Iron and ATP Production
ATP (adenosine triphosphate) serves as your body’s energy currency, and iron plays a critical role in its production. Inside your mitochondria, iron-containing proteins like cytochromes shuttle electrons along the electron transport chain, creating the electrochemical gradient that powers ATP synthesis. This process is remarkably efficient but absolutely dependent on adequate iron levels.
3. Shortness of Breath During Normal Activities
Finding yourself winded after climbing a flight of stairs or carrying groceries when you used to handle these tasks with ease? This could be iron deficiency speaking. When your hemoglobin levels drop, your body struggles to deliver sufficient oxygen to your tissues, forcing your respiratory and cardiovascular systems to work overtime to compensate.
Your heart rate increases and your breathing becomes more rapid as your body attempts to circulate the limited oxygen-carrying red blood cells more quickly. This compensation mechanism explains why even mild exertion can leave you breathless when your iron stores are depleted. The effect becomes particularly noticeable during physical activity but can manifest even during routine daily tasks.
4. Headaches and Dizziness
- Recurring headaches with no other obvious cause
- Lightheadedness when standing up quickly
- A feeling of brain fog or difficulty concentrating
- Occasional vertigo or feeling that the room is spinning
These neurological symptoms directly connect to oxygen delivery problems in your brain. Your brain consumes approximately 20% of your body’s oxygen supply despite comprising only 2% of your body weight. When iron levels drop, the brain experiences the oxygen shortage acutely, triggering these uncomfortable symptoms.
Many people mistake these symptoms for stress, dehydration, or lack of sleep, allowing iron deficiency to progress unchecked. The discomfort typically begins subtly but becomes more persistent and pronounced as iron stores continue to deplete.
If you experience these symptoms alongside other signs of iron deficiency, consider testing your iron levels before assuming another cause. Simple blood tests measuring ferritin (your iron storage protein) can reveal whether iron deficiency might be behind your headaches and dizzy spells.
5. Unusual Cravings for Ice or Dirt
One of the most curious and distinctive symptoms of iron deficiency is a condition called pica—the craving and consumption of non-food substances. The most common manifestation is an inexplicable urge to chew ice (pagophagia), but some people develop cravings for dirt, clay, paper, or other unusual substances. Scientists haven’t fully explained the connection, but the relationship between severe iron deficiency and these peculiar cravings is well-documented.
If you’ve found yourself mindlessly crunching through cups of ice or experiencing unusual cravings, your body might be signaling a serious iron shortfall. While these symptoms sound bizarre, they’re actually recognized medical indicators of iron deficiency anemia. The good news is that these unusual cravings typically resolve once iron levels return to normal.
Who Needs More Iron? High-Risk Groups
While anyone can develop iron deficiency, certain populations face significantly higher risk due to biological needs, physiological changes, or dietary patterns. Understanding these risk factors can help you assess your own likelihood of developing low iron status and take preventive action before symptoms develop.
Women During Menstruation and Pregnancy
Women of reproductive age face a double challenge with iron. Monthly menstrual blood loss creates a recurring iron drain that can deplete stores over time, especially with heavy periods. The average menstruating woman loses about 30-40 ml of blood monthly, which equals approximately 0.5-0.7 mg of iron lost daily when averaged across the month—nearly double the iron loss experienced by men.
Pregnancy dramatically increases iron requirements as maternal blood volume expands by almost 50% and the developing fetus and placenta consume additional iron. A pregnant woman needs approximately 27 mg of iron daily compared to the 18 mg recommended for non-pregnant women. This increased demand often cannot be met through diet alone, which is why prenatal supplements containing iron are typically recommended. For more information on essential nutrients, you can explore the importance of copper nutrition.
Post-childbirth blood loss further depletes iron stores, and breastfeeding requires continued attention to iron status. Women who experience multiple pregnancies close together face compound challenges in maintaining healthy iron levels, as their bodies have limited time to replenish stores between pregnancies.
Growing Children and Teenagers
Children and adolescents require substantial iron to support their rapid growth, expanding blood volume, and developing tissues. Teenage girls face a particularly high risk once menstruation begins, creating a perfect storm of increased needs and ongoing losses. During growth spurts, a teenager’s iron requirements can nearly double, yet many consume diets low in bioavailable iron sources.
Athletes and Highly Active People
Exercise increases iron needs through multiple mechanisms. Intense physical activity accelerates red blood cell production, expands total blood volume, and increases the density of energy-producing mitochondria—all processes requiring additional iron. Regular impact exercise can also cause microscopic bleeding in the digestive tract and increase iron loss through sweat, further increasing requirements.
Endurance athletes face a particularly high risk, with studies showing up to 70% of female distance runners may have suboptimal iron levels. The combination of high training volumes, increased iron turnover, and sometimes restricted eating patterns creates a perfect storm for iron depletion. Male athletes aren’t immune either—while they start with higher baseline iron stores, intensive training still increases their requirements significantly. For more information on another essential mineral, you can explore the benefits of cobalt nutrition.
Even recreational exercisers should monitor their iron status if they increase training intensity or volume significantly. The body’s initial adaptation to increased exercise often includes expanding blood volume and producing more red blood cells—both processes that require substantial iron. Without adequate intake, these adaptations may be limited, hampering performance improvements.
Exercise-Induced Iron Losses
• Foot-strike hemolysis: Red blood cell destruction from repetitive impact
• Gastrointestinal bleeding: Micro-bleeding in the digestive tract during prolonged exercise
• Sweat losses: 0.3-0.4 mg iron lost per liter of sweat
• Increased turnover: Accelerated red blood cell recycling with training
Vegetarians and Vegans
Plant-based eaters face two significant iron-related challenges: their diets provide only non-heme iron (which has lower bioavailability) and often contain higher levels of compounds that inhibit iron absorption. While well-planned vegetarian and vegan diets can provide sufficient iron, they typically require more thoughtful food combinations and sometimes 1.8 times more total iron intake to compensate for reduced absorption rates. For those interested in other essential nutrients, exploring copper nutrition can also be beneficial in maintaining a balanced diet.
Top Iron-Rich Foods to Add to Your Diet
Addressing iron needs through diet requires understanding not just which foods contain iron, but which forms are most bioavailable and how to enhance absorption. The foods you choose and how you combine them can dramatically impact how much iron your body actually absorbs.
Iron exists in two dietary forms: heme iron from animal sources and non-heme iron from plant sources. Your body absorbs heme iron at a rate of 15-35%, while non-heme iron absorption ranges from just 2-20%, depending on your iron status and the presence of absorption enhancers or inhibitors in your meal. For those interested in other essential minerals, you might want to explore the importance of copper nutrition as well.
Animal-Based Iron Champions

Animal foods provide the most readily absorbed form of iron (heme iron), making them efficient choices for improving iron status. Organ meats like liver contain the highest concentrations, with a single 3-ounce serving of beef liver providing over 5 mg of highly bioavailable iron. Regular consumption of even small amounts of organ meats can significantly impact iron status.
Beyond organ meats, red meat (especially beef) delivers substantial iron, with 3 ounces of lean beef providing approximately 2.5 mg. Interestingly, darker meat from poultry (like chicken thighs) contains more iron than white meat, and certain seafoods—particularly oysters, clams, and mussels—rank among the most iron-rich foods available. These shellfish combine excellent iron content with heart-healthy omega-3 fatty acids, making them nutritional powerhouses.
Plant-Powered Iron Sources

While plant foods contain only non-heme iron with lower absorption rates, strategic combinations can help vegetarians and vegans meet their iron needs. Legumes like lentils, chickpeas, and beans offer impressive iron content, with a cup of lentils providing around 6.6 mg. Dark leafy greens such as spinach and kale contribute significant iron while delivering additional nutrients like folate and vitamin C that support overall blood health.
Whole grains, especially quinoa and amaranth, contain more iron than refined grains because the mineral concentrates in the bran and germ removed during processing. Tofu and tempeh also deserve special mention for their dual contribution of iron and complete protein. For convenient iron-rich snacks, pumpkin seeds, sesame seeds, and dried apricots make excellent choices that can easily be incorporated into daily eating patterns.
Iron Absorption Enhancers to Pair With Your Meals

- Vitamin C-rich foods (citrus fruits, bell peppers, strawberries) can increase non-heme iron absorption by up to 300%
- Fermented foods like sauerkraut and kimchi help break down phytates that inhibit iron absorption
- Carotenoid-rich foods such as carrots and sweet potatoes may enhance iron absorption
- Using acidic marinades containing lemon juice or vinegar when preparing iron-rich foods
- Garlic and onions contain sulfur compounds that may promote iron availability
Thoughtful food pairings can dramatically increase the iron you absorb from each meal. For instance, adding sliced bell peppers to a spinach salad or squeezing lemon over lentil soup can significantly enhance iron absorption. Similarly, combining iron-rich foods with those containing vitamin A and beta-carotene appears to help overcome the inhibitory effects of phytates and polyphenols. For more information on how to improve your iron intake, check out this comprehensive guide on iron absorption.
Iron Supplements: When and How to Take Them
While a well-planned diet should be your first approach to maintaining iron levels, supplements sometimes become necessary. Iron deficiency is the most common nutritional deficiency worldwide, and when diet alone cannot correct low iron status, supplementation provides an effective intervention. However, iron supplements aren’t appropriate for everyone and require careful consideration of form, timing, and potential interactions.
Types of Iron Supplements
Iron supplements come in various forms, each with different absorption rates and side effect profiles. Ferrous sulfate represents the most common and inexpensive option, typically providing 65 mg of elemental iron per 325 mg tablet. Ferrous gluconate and ferrous fumarate offer gentler alternatives that may cause less digestive distress, though they provide slightly less elemental iron per dose.
For those experiencing significant digestive discomfort with standard supplements, newer options include iron bisglycinate (iron bound to amino acids) and micronized, dispersible ferric pyrophosphate. These formulations often cause fewer gastrointestinal side effects while maintaining good bioavailability. Liquid iron supplements or slow-release formulations provide additional options for those unable to tolerate standard iron pills, though absorption rates may vary. For more information on related minerals, you can explore the benefits of copper nutrition.
Proper Timing for Maximum Absorption
Timing significantly impacts how much iron your body absorbs from supplements. Taking iron on an empty stomach generally maximizes absorption, with studies showing up to three times greater absorption compared to taking it with meals. However, this approach often increases digestive side effects, so finding your personal balance between maximum absorption and minimum discomfort may require experimentation.
If you need to take iron with food to reduce stomach upset, avoid combining it with calcium-rich foods, whole grains, coffee, tea, or dairy, as these significantly impair absorption. Instead, pair your supplement with a small amount of vitamin C-rich food to enhance absorption while minimizing digestive distress. For optimal results, separate iron supplements from other medications by at least two hours, particularly antacids, calcium supplements, thyroid medications, and certain antibiotics.
Common Side Effects and How to Minimize Them
Iron supplements frequently cause digestive discomfort, including constipation, nausea, stomach pain, and in some cases, diarrhea. These side effects result primarily from unabsorbed iron irritating the digestive tract. Starting with a lower dose and gradually increasing it can help your system adjust and reduce these effects. Taking supplements with small amounts of food, despite slightly reduced absorption, often provides a reasonable compromise between effectiveness and comfort.
Constipation represents the most common complaint with iron supplementation. Increasing water intake, consuming more fiber-rich foods, and mild physical activity can help mitigate this effect. If these approaches prove insufficient, your healthcare provider might recommend a stool softener or suggest switching to a different iron formulation. Remember that iron supplements temporarily darken stools—this harmless effect simply indicates unabsorbed iron and shouldn’t cause concern.
Too Much of a Good Thing: Iron Overload Risks
While iron deficiency receives considerable attention, excessive iron carries its own significant risks. Iron stands alone among minerals in that the body lacks an efficient excretion mechanism for removing excess amounts. Once absorbed, iron remains in your system until used or lost through bleeding, cell sloughing, or other minor pathways. This limited elimination capability means excess iron accumulates in tissues over time, potentially causing serious damage.
Free iron particles can generate harmful free radicals through a process called the Fenton reaction. These reactive oxygen species damage cellular structures including proteins, lipids, and DNA, accelerating aging and increasing disease risk. Organs particularly vulnerable to iron accumulation include the liver, heart, pancreas, and brain—explaining why iron overload often manifests as liver disease, heart problems, diabetes, and neurological symptoms.
Hemochromatosis: When Your Body Stores Too Much Iron
Hereditary hemochromatosis represents the most common genetic cause of iron overload, affecting approximately 1 in 300 people of Northern European descent. This condition results from mutations in genes controlling iron absorption, causing the body to continue absorbing iron despite adequate or excessive stores. Left untreated, hemochromatosis leads to progressive iron accumulation in vital organs, potentially causing cirrhosis, heart failure, diabetes, arthritis, and increased cancer risk.
Early symptoms of hemochromatosis often appear vague and nonspecific—fatigue, joint pain, abdominal discomfort—making diagnosis challenging without appropriate testing. The condition frequently goes unrecognized until organ damage has occurred, highlighting the importance of genetic testing for those with family history and routine iron screening for at-risk populations. Fortunately, when identified early, regular therapeutic phlebotomy (blood removal) effectively manages hemochromatosis by reducing iron stores.
Warning Signs of Excessive Iron
Signs of iron overload often develop gradually and can mimic many other conditions. Early symptoms typically include unexplained fatigue, joint pain (particularly in the knuckles of the first and second fingers), abdominal discomfort, and decreased libido. As iron accumulation progresses, skin darkening (bronze diabetes), irregular heart rhythms, liver enlargement, and diabetes may develop. Anyone experiencing these symptoms, especially with a family history of hemochromatosis or unexplained liver disease, should discuss iron testing with their healthcare provider.
Boost Your Energy With These Iron-Focused Habits
Optimizing your iron status requires more than simply consuming iron-rich foods or supplements. Strategic habits that enhance absorption while avoiding common pitfalls can dramatically improve your iron balance and energy levels. Cooking acidic foods in cast iron cookware can increase iron content by 16-37%, especially for acidic recipes like tomato sauce. Try spacing iron-rich meals throughout the day rather than concentrating them at one meal, which saturates absorption pathways. When taking supplements, alternating days may prove more effective than daily dosing for some people, as absorption increases when iron stores drop slightly between doses. For athletes, periodizing iron intake to align with training cycles helps support adaptation while minimizing oxidative stress during recovery periods.
Iron Infographic
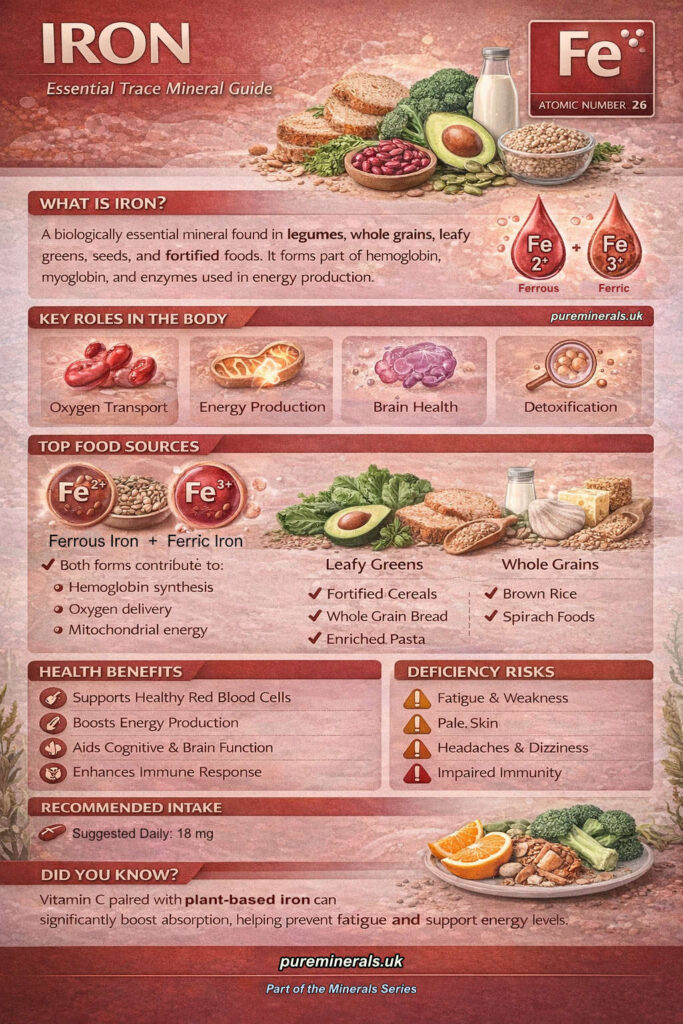
Download the full high-resolution Iron infographic for offline reference.
Explore more minerals in the full Minerals Hub
Frequently Asked Questions
The relationship between iron, oxygen transport, and energy production generates many questions. Here are answers to the most common inquiries about optimizing iron status and function in your body.
How much iron do I need daily?
Daily iron requirements vary significantly based on age, sex, and life stage. Adult men and postmenopausal women generally need 8mg daily, while menstruating women require 18mg to compensate for menstrual losses. Pregnant women need significantly more—27mg daily—to support expanded blood volume and fetal development. Athletes, especially those in endurance sports, may require up to 30% more iron than sedentary individuals due to increased turnover and losses through sweat. Remember that these recommendations reflect total intake needed, not accounting for absorption differences between heme and non-heme sources.
Can drinking tea or coffee affect my iron levels?
Yes, both tea and coffee can substantially reduce iron absorption when consumed with meals. The polyphenolic compounds (particularly tannins) in these beverages bind to iron, forming insoluble complexes that your body cannot absorb. Studies show that tea can reduce iron absorption by up to 60-70%, while coffee may decrease it by 35-50%. This effect applies primarily to non-heme iron from plant sources, with less impact on heme iron from animal foods.
Beverage | Potential Reduction in Iron Absorption | Recommendation |
|---|---|---|
Black Tea | 60-70% | Separate from meals by 1-2 hours |
Coffee | 35-50% | Separate from meals by 1 hour |
Green Tea | 25-50% | Separate from meals by 1 hour |
Herbal Tea (non-caffeinated) | 0-20% (varies by herb) | Most can be consumed with meals |
To minimize this effect without giving up your favorite beverages, simply separate tea and coffee consumption from iron-rich meals and supplements by at least one hour. Many people find consuming these beverages between meals rather than with them provides an easy solution. Adding milk to tea may actually reduce its iron-inhibiting effects by binding some of the tannins, while adding lemon to tea increases vitamin C, potentially offsetting some of the inhibitory effects.
If you’re actively working to improve low iron status, consider temporarily reducing your tea and coffee consumption or being particularly strict about separating them from iron-rich meals and supplements. Once your iron levels normalize, you can typically return to normal consumption patterns while maintaining awareness of timing.
Is cooking in cast-iron pans a good way to increase iron intake?
Cooking in cast iron cookware does indeed increase the iron content of food, particularly when preparing acidic recipes that remain in the pan for extended periods. Research shows that tomato sauce cooked in cast iron can increase its iron content by over 500%, while more neutral foods like cornbread might see increases of 20-30%. The effect proves most pronounced with acidic foods (tomatoes, citrus, vinegar), longer cooking times, higher moisture content, and newer or less seasoned pans. While this source of additional iron won’t correct severe deficiency on its own, it provides a practical strategy to complement dietary sources, especially for those at risk of mild deficiency.
How long does it take to correct an iron deficiency?
The timeline for correcting iron deficiency varies considerably based on its severity, the treatment approach, and individual factors affecting absorption and utilization. With proper supplementation, hemoglobin levels typically begin improving within 2-4 weeks, though you may notice energy improvements sooner as tissue enzymes receive more iron. Complete replenishment of iron stores (measured by ferritin) takes considerably longer—often 3-6 months of consistent supplementation. Those with severe deficiency or ongoing losses may require 6-12 months to fully restore iron reserves. Regular monitoring through blood tests helps track progress and adjust treatment as needed, preventing both under-correction and iron overload. Understanding other minerals, like copper, that affect iron absorption can also be beneficial.
Can certain medications interfere with iron absorption?
Several common medications significantly impact iron absorption and should be separated from iron-rich meals or supplements. Antacids and acid reducers (H2 blockers, proton pump inhibitors) decrease stomach acid, which normally helps convert dietary iron to its absorbable form. Calcium supplements and calcium-containing antacids can reduce iron absorption by up to 60% when taken simultaneously. Certain antibiotics, particularly tetracyclines and quinolones, form chelates with iron that prevent absorption of both the antibiotic and the iron. For those taking these medications, careful timing—generally separating iron intake by 2-4 hours—helps maximize the effectiveness of both the medication and iron absorption.
The relationship between iron and your body’s oxygen transport system fundamentally determines your energy levels, cognitive function, and overall vitality. By understanding this mineral’s critical role and implementing strategies to optimize your iron status, you can support everything from daily energy to athletic performance and long-term health. Remember that balance remains key—both deficiency and excess carry significant consequences.
